NEWS

iLine Microsystems has put its stakes on an Industry 4.0 strategy to meet the requirements of a globalised market.
iLine Microsystems, a Gipuzkoa-based biotechnology firm specialised in developing in-vitro diagnostic systems used to monitor oral anti-clotting therapies, has decided to implement an Industry 4.0 strategy to meet the needs of a complex biohealth sector.
This highly regulated market is based on life cycles that are becoming increasingly shorter. This requires that the entire value chain be integrated, especially in terms of after-sales services. It is also demanding enhanced product performance based on increasing the rate of valid tests relative to rejects.
Integrating and managing processes efficiently and introducing ICTs to facilitate real-time decision-making are some of the key elements needed to set up a competitive production model in those markets in which iLine Microsystems is currently involved.
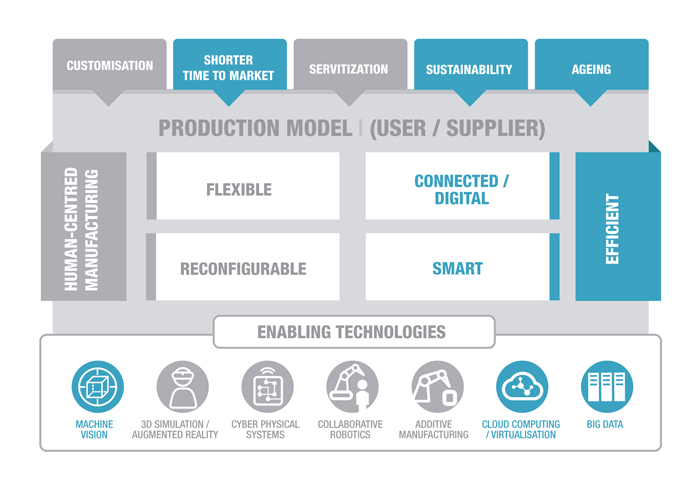
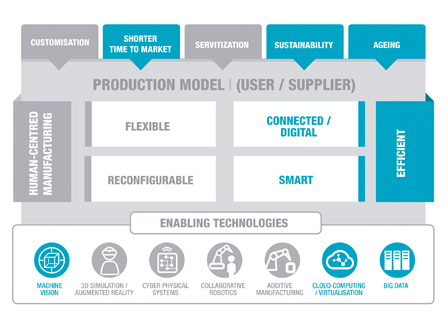
The key elements of the business and the technological contribution made by IK4-TEKNIKER
The solution designed by iLine Microsystems that measures clotting times represents a paradigm of efficiency as regards bringing together heterogeneous technologies such as photonics, microfluidics, micromechanics, signal processing, algorithmics and biochemistry. IK4-TEKNIKER is collaborating with iLine Microsystems to develop this concept of technological integration and, amongst other things, to design and implement a viewing system the measuring principle is based on.
The product available on the market is a kit made up of a portable analyser and disposable analytical strips (microfluidic chip). It must be highlighted that one of the strengths of the company is connected to total production control. The chip is mass-produced (thousands of units every day) at the San Sebastian plant in accordance with the highest safety standards.
The key underlining elements of the iLine Microsystems production system applied to meet the requirements of this highly regulated, growing and globalised market are, amongst others, total traceability of a product and its associated processes together with efficiency throughout the entire life cycle. In this regard, co-operation with the technology centre also covers stages of verification, validation, certification and after-sales services of iLine Microsystems products.
In this respect, and in order to deliver a suitable process, a strategic approach is required to properly manage the vast amount of information generated during a product’s life cycle. Heterogeneous systems of this kind require partial integrations allowing for significant conclusions to be drawn in terms of performance and reliability so that dependencies between modules can be established.
Proper management of all this information requires a suitable architecture and advanced information systems to achieve total traceability with regard to the product development process, production, distribution and use.
It is also necessary to apply advanced analytical solutions to allow for the use of information originating from device utilisation in the form of feedback aimed at the product improvement process. Finally, it must be possible to test and certify any modifications on components and assess their impact on the end product.